|
x |
x |
|
 |
 |
|
INFECTIOUS
DISEASE |
BACTERIOLOGY |
IMMUNOLOGY |
MYCOLOGY |
PARASITOLOGY |
VIROLOGY |
|
TURKISH |
VIROLOGY - CHAPTER TWENTY ONE
ARBOVIRUSES
Dr Margaret Hunt
Professor Emerita
University of South Carolina School of Medicine
Columbia
South Carolina
|
|
Español |
|
|
Let us know what you think
FEEDBACK |
|
SEARCH |
|
|

 |
|
|
|
|
|
TEACHING
OBJECTIVES
Introduction
to viral zoonoses
Brief overview of general features of togaviruses, bunyaviruses, reoviruses, arenaviruses and flaviviruses
Discussion of
ecology, epidemiology and public health
Arbovirus encephalitis, febrile and hemorrhagic
disease
Rodent borne
hemorrhagic fever, hemorrhagic fever with renal syndrome and hantavirus
pulmonary syndrome.
Flavivirus-associated hemorrhagic fevers |
INTRODUCTION
Zoonotic viruses are viruses which are transmissible from animals (arthropods,
vertebrates) to man. Many are transmitted by means of an infected,
blood-sucking, arthropod vector (arthropod borne =
arboviruses). Others may be transmitted by inhalation, or conjunctival contact
with infected excretions, or by direct contact with infected animal (e.g.
rabies).
Constant vigilance and surveillance are important
components in reducing the public health impact of these viruses
|
ARBOVIRUSES |
|
FAMILY
|
ENVELOPE
|
SYMMETRY
|
GENOME
|
IMAGE
|
SIZE* |
|
Togaviridae
Flaviviridae
|
yes
|
icosahedral
|
single strand RNA (+ve)
|
 Computer generated model of the surface of an alphavirus derived by
cryoelectron microscopy. CDC
Computer generated model of the surface of an alphavirus derived by
cryoelectron microscopy. CDC
|
 |
|
Bunyaviridae
|
yes
|
helical
|
single strand RNA (-ve) segmented
|
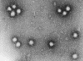 Copyright The Australian National University
Copyright The Australian National University
|
 |
|
Reoviridae
|
no
|
icosahedral
|
double strand RNA segmented
|

Transmission electron micrograph of reovirus type 3.
Both inner and outer capsid shells are present. Some
virions are penetrated by negative stain.
CDC/Dr. Erskine Palmer
|
 |
|
* Relative size adapted from White and Fenner , Medical Virology, 1994 |
The term arboviruses is used to describe viruses from various
families which are transmitted via arthropods. Diseases caused by arboviruses
include encephalitis, febrile diseases (sometimes with an associated rash), and
hemorrhagic fevers.
|
 Components
in the transmission and maintenance of Arboviral Encephalitis
CDC
Components
in the transmission and maintenance of Arboviral Encephalitis
CDC |
ARTHROPOD VECTORS
The virus replicates in
the vector but usually does not harm it. In the mosquito-borne diseases, the
virus establishes a persistent infection in the salivary glands and there is
sufficient virus in the saliva to infect another host during feeding. Each virus
usually only grows in a limited number of mosquito species.
The natural habitat of the
vector (rural or around dwellings), its diurnal activity patterns and its preferred
targets for a blood meal, affect the transmission pattern of the disease. Many
vectors are only active during part of the year and so this will affect the seasonal
incidence of the disease. Insect vectors may over-winter as eggs; in this case
the virus must either be transmitted transovarially (which happens in some
cases), or survive the
winter in the vertebrate host and infect the vector the following year.
VERTEBRATE HOSTS
The virus is usually
maintained in a vertebrate host. There is often a persistent viremia and the
host can act as a long term reservoir. In many cases the reservoir host is not
severely affected by the viral infection. If the vertebrate reservoir is
migratory, this will affect the timing of infections in a particular locale.
DEAD END HOSTS
In many cases, if the
virus is transmitted to an animal other than its normal host, viremia is low or
transient and there is little chance of the infected animal being able to pass
enough virus to a blood sucking arthropod to establish an infection. In this
case, the animal is said to be a dead-end host. Man is a dead-end host in the
case of most arbovirus diseases (exceptions include yellow fever and dengue fever).
PREVENTION
Methods include surveillance, vector control,
public education about reducing breeding sites for vectors and reducing exposure
to vectors (by wearing suitable clothing, using insect repellents, timing
activities for low risk time of day, etc.), and use of vaccines in few cases
where available.
|
 Sylvatic life cycle
Sylvatic life cycle
 Urban life cycle
Urban life cycle |
LIFE CYCLES OF ARBOVIRUSES
There are several types of
life cycles, but many arboviruses have a sylvatic cycle while some also have an urban
cycle.
1. Sylvatic cycle (sometimes
known as the jungle cycle). In this the virus cycles between an arthropod and a
mammalian host with man usually a dead-end host infected by the arthropod
2. Urban cycle. In this the
virus cycles between man and an arthropod species.
There is an urban cycle
for yellow fever and dengue fever (both of these also have
a sylvatic/jungle cycle). If there is an urban cycle, using window
screens, bed nets, etc. to prevent access of mosquitos to viremic patients may
reduce transmission.
ARBOVIRUS DISEASE
Diseases caused by arboviruses include:
There is some overlap of symptoms between various virus types; for example viruses not usually classed as encephalitis viruses may cause CNS symptoms, etc.
More than 100 arboviruses are known to infect man but only a few will be mentioned here. Disease outbreaks caused by arboviruses are sporadic and unpredictable.
Usually, infection is followed by replication in endothelial cells and
macrophage/monocyte lineage cells. Frequently, these infections are associated with flu-like
symptoms since these RNA viruses are good inducers on interferon. Often the infection stops here but it may produce enough virus for secondary
viremia which can then cause a major infection of target organs (brain,
endothelial cells, liver). Which organs are targeted depends on the tropism of
the virus. Access to the brain tends to be via infection of the endothelial cells in the blood vessels supplying the
brain.
RECOVERY
Recovery involves the cell-mediated immune system. The
arboviruses are generally good inducers of interferon, which may partially
explain early influenza-like symptoms common to so many of these viruses (fever,
headache, fatigue,
myalgia).
Antibody can be important in controlling the secondary viremia and limiting
disease.
DIAGNOSIS
Diagnosis is difficult because many other agents cause similar symptoms. Arbovirus infection is usually confirmed by immunological methods (complement
fixation, ELISA, immune fluorescence assay, etc) or by
PCR of the viral nucleic acid. The tests are usually done in a State Laboratory or at the Centers for Disease Control.
Awareness of an arbovirus associated disease in a particular area enables risk reduction procedures to be put in place (vector-control, insect repellents, protective
clothing, change in human activity patterns).
RESISTANCE
Resistance to arboviruses is mediated by antibodies and recovery
involves the cell-mediated immune system. |
| |
ARBOVIRUS ENCEPHALITIS
DISEASES
|
ARBOVIRUSES - ENCEPHALITIS
- Viruses currently
important in the USA |
|
NAME
|
DISEASE
|
OCCURRENCE
|
VECTOR
|
RESERVOIR |
|
Flaviviridae Family
|
|
St Louis Encephalitis Virus
|
encephalitis
|
North America
|
Mosquito

|
Birds |
|
West Nile Virus
|
encephalitis
|
East and Central North America, parts of Europe
and Africa
|
Mosquito
|
Birds |
|
Bunyaviridae Family
|
|
La Cross Virus
(California serogroup)
|
encephalitis
|
North America
|
Mosquito
|
Small mammals |
|
Togaviridae Family
|
|
Eastern Equine Encephalitis Virus
|
encephalitis
|
Eastern U.S., Canada
|
Mosquito
|
Birds |
|
Western Equine Encephalitis Virus
|
encephalitis
|
Western U.S., Canada, Mexico, Brazil
|
Mosquito
|
Birds |
|
Venezuelan Equine Encephalitis
|
encephalitis
|
Central and South America, Texas, Florida
|
Mosquito
|
Small mammals
Horses |
All of the arboviruses in the
above table are
transmitted by mosquitoes; however, some arbovirus encephalitides are transmitted
by ticks or other insect vectors such as sandflies. For all of the viruses listed
below,
most infections are sub-clinical, but if clinical cases do occur, the
consequences may be very serious. Initial symptoms are flu-like including fever, but can
progress to encephalitis. The following are the most frequently
reported in humans in United States
|
|
La Crosse Encephalitis
/ California serogroup
La Crosse Virus - Bunyavirus family
The reservoir for La Crosse virus is small
mammals (chipmunks and squirrels) and the virus is transmitted by mosquitoes (Aedes
triseriatus, Easter Treehole mosquito). Humans are "dead end" hosts as they
do not develop a sufficiently high viremia to reinfect a mosquito.
The incubation period is one
to two weeks after a bite by an infected mosquito.
Symptoms include:
-
fever
-
headache
-
nausea
-
lethargy
Children more often develop symptoms than
adults and are more prone to neuroinvasive disease (resulting in seizures) but morbidity and fatality are low (case fatality rate is less than
1%). Severe neuroinvasive disease may result in repeated seizures, partial
paralysis and neurobehavioral changes.
Recent cases have been predominantly in the Eastern United States
where there are about 80 La Crosse virus neuroinvasive cases per year.
There is no vaccine and treatment is supporive including seizure
control.
|
|
 Negatively-stained transmission electron micrograph showing the presence of La
Crosse encephalitis virus ribonucleoprotein particles. CDC/ Dr. J. Obijeski
Negatively-stained transmission electron micrograph showing the presence of La
Crosse encephalitis virus ribonucleoprotein particles. CDC/ Dr. J. Obijeski |
 A
A
 B
B
California Serogroup Virus Neuroinvasive Disease* Cases Reported by
Year, A: 1964-2010 B: 2004-2013
CDC
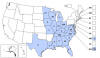 California Serogroup Virus. Neuroinvasive Disease Cases Reported by
State, 2004-2013 CDC
California Serogroup Virus. Neuroinvasive Disease Cases Reported by
State, 2004-2013 CDC
|
 St Louis encephalitis virus transmission
cycle CDC
St Louis encephalitis virus transmission
cycle CDC
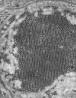 Negatively-stained transmission electron micrograph (TEM) showing the presence
of numerous St. Louis encephalitis virions that were contained within a mosquito
salivary gland tissue sample. CDC/ Dr. Fred Murphy; Sylvia Whitfield
Negatively-stained transmission electron micrograph (TEM) showing the presence
of numerous St. Louis encephalitis virions that were contained within a mosquito
salivary gland tissue sample. CDC/ Dr. Fred Murphy; Sylvia Whitfield
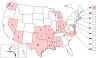 St. Louis Encephalitis Virus Neuroinvasive Disease Cases* Reported by
State, 2004-2013 CDC
St. Louis Encephalitis Virus Neuroinvasive Disease Cases* Reported by
State, 2004-2013 CDC |
St. Louis Encephalitis
St. Louis Encephalitis virus - Flavivirus family
The elderly are most severely affected by a St. Louis
Encephalitis Virus
(SLEV) infection since they often have weaker immune systems. The case fatality rate varies from 3-25%. The reservoir is birds and the virus
is transmitted by mosquitoes with humans as a "dead end host". This virus can have an urban cycle as well as well
as a sylvatic cycle. Infections occur in the northern parts of the United States
in the late summer and early fall but in the south, infections occur all year.
The geographic range of this virus is most of North and South America but
most cases of human disease have occurred in the United States. The most
extensive epidemic of SLEV infections occurred in 1975 in the
Ohio-Mississippi river basin.
In many cases of SLEV infection, mild infections occur
without apparent symptoms other than fever and headache. In more severe
cases, there are:
The
symptoms are therefore similar to West Nile encephalitis which is also caused by
a flavivirus (see below). The number of cases seen in the United States varies
widely with more than 100 annually but only 1 to 12 cases of St Louis
Encephalitis Virus neuroinvasive disease reported between 2004 and 2013.
There is no vaccine for this virus for use in humans but
infection seems to cause life-long immunity.
|
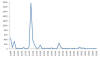 A
A
 B
B
St. Louis Encephalitis Virus Neuroinvasive Disease Cases*
Reported by Year, A: 1964-2010. Note peak in 1975 B: 2004-2013.
CDC
|
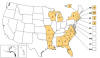
 St. Louis Encephalitis Virus Neuroinvasive Disease* Average Annual
Incidence by County, 1996-2010
St. Louis Encephalitis Virus Neuroinvasive Disease* Average Annual
Incidence by County, 1996-2010
* Neuroinvasive disease includes cases reported as
encephalitis, meningoencephalitis, or meningitis. CDC
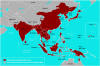 The geographic distribution of the Japanese encephalitis serocomplex of the
family Flaviridae, 2016 CDC
The geographic distribution of the Japanese encephalitis serocomplex of the
family Flaviridae, 2016 CDC
|
 A
A
 B
B
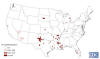
Eastern Equine Encephalitis Virus Neuroinvasive Disease
Cases Reported by Year. A: 1964-2010 B: 2004-2013 CDC
 Eastern Equine Encephalitis Virus Neuroinvasive Disease Cases Reported
by State, 2004--2013
Eastern Equine Encephalitis Virus Neuroinvasive Disease Cases Reported
by State, 2004--2013
CDC
|
Eastern Equine
Encephalitis
Eastern Equine Encephalitis Virus - Alphavirus genus of Togavirus family
The reservoir of
Eastern Equine Encephalitis virus (EEEV) is birds and it is transmitted by
mosquitoes. Culiseta melanura mosquitoes transit the virus
between birds and a variety of mosquito species transmit the virus
from birds to humans and other mammals. Humans and horses are "dead end hosts". Many cases result in mild
flu-like symptoms or no symptoms at all but children are more likely to have severe clinical symptoms that adults.
The incubation period is about 4 to 10 days. People under 15 or older than 50 are more likely to have severe
clinical symptoms. Disease that does not involve the brain is systemic
and results in:
- chills, fever, muscle and joint pain and a general feeling
of malaise
The symptoms resolve in a few weeks
The encephalitic form, involving the central nervous system, is
somewhat different according to age. In infants, the disease is
manifested abruptly while older children exhibit symptoms of
systemic disease first. Symptoms of encephalitic disease include:
- Headache
- Fever
- sleepiness
- Irritability
- anorexia
- nausea
- diarrhea
- cyanosis
- convulsions
- coma
If there is
clinical disease, death may occur relatively frequently (Case fatality rate is about 35%). Sequelae
(mild to severe neurologic deficits) are
common in survivors. The disease is quite rare and there have been about 250 confirmed human cases of Eastern Equine
encephalitis in the United States since 1964. Mostly EEEV disease is
seen in the Atlantic and Gulf Coast states.
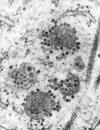 Transmission electron micrograph showing the presence of a number of
Eastern Equine Encephalitis (EEE) virus virions that happened to be
in a specimen of central nervous system tissue. CDC/ Dr. Fred
Murphy; Sylvia Whitfield
Transmission electron micrograph showing the presence of a number of
Eastern Equine Encephalitis (EEE) virus virions that happened to be
in a specimen of central nervous system tissue. CDC/ Dr. Fred
Murphy; Sylvia Whitfield
Western Equine Encephalitis
Western Equine Encephalitis Virus - Alphavirus genus of Togavirus family
Again the reservoir is birds and the virus is transmitted by mosquitoes. Humans
and horses are a dead end hosts. As with eastern equine encephalitis, children
are more likely to have severe clinical symptoms that adults. The case fatality
rate is 3-7%. In the young,
death or sequelae (mild to severe neurological impairment) are more common.
There have been 639 confirmed cases in the U.S. since 1964; however the number
has dropped off in recent years.
Venezuelan Equine
Encephalitis
Venezuelan Equine Encephalitis
- Alphavirus genus of Togavirus
family
This virus is predominantly a problem in central and South
America, but it periodically occurs in the southern United States (Texas,
Florida). Its reservoir is small mammals and horses.
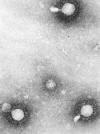 Negative stain electron
micrograph showing the presence of a number of Venezuelan equine
encephalitis virus virions. CDC/ Dr. Fred Murphy; Sylvia Whitfield
Negative stain electron
micrograph showing the presence of a number of Venezuelan equine
encephalitis virus virions. CDC/ Dr. Fred Murphy; Sylvia Whitfield
|
|
WEB RESOURCES
St Louis Encephalitis
CDC
Information - St Louis Encephalitis
Western Equine Encephalitis
CDC
information |
|
CASE REPORT
Cases of Eastern
Equine Encephalitis in the Young and the Old |
| |
|
|
West Nile Encephalitis
West Nile Encephalitis Virus - Flavivirus
family
West Nile virus is
closely related to St. Louis encephalitis virus and until 1999 was
found in Africa, west Asia/eastern Europe and the Middle East. In 1999
it was detected in North America, initially on the east coast, and
has subsequently spread across both the United States and Canada.
Most
(75%) of people who become infected show no symptoms and mount a
successful immune attack against the virus. Most of the remainder (1
in 5 infected persons) have
flu-like symptoms (fever, headache and general malaise) with an
incubation period of 3 to 14 days. Sometimes
there is swelling of the lymph glands (lymphadenopathy) and there may also be a rash. In
less than 1% of patients, the infection is life-threatening as a
result of encephalitis or meningitis. The initial symptoms are:
-
high fever
-
headache
-
stiff neck
These are followed by:
Recovery can take
weeks or months. Persons over 50 years
of age are most severely affected by West Nile encephalitis and the case fatality rate
is
approximately 10%.
People with certain diseases of
immunocompromization and those with cancer, diabetes, kidney disease
and hypertension are more at risk for overt disease.
Some people who become
infected with West Nile Virus can develop “acute flaccid paralysis”.
This is a sudden onset of weakness in the limbs and/or breathing
muscles. In most persons, this syndrome is due to the development of
West Nile poliomyelitis—an inflammation of the spinal cord that causes
a syndrome similar to that caused by the poliovirus. West Nile
poliomyelitis was first widely recognized in the United States in
2002. Persons with West Nile poliomyelitis may develop sudden or
rapidly progressing weakness. The weakness tends to affect one side of
the body more than the other, and may involve only one limb. The
weakness is generally not associated with any numbness or loss of
sensation, but may be associated with severe pain. In very severe
cases, the nerves going to the muscles that control breathing may be
affected, resulting in rapid onset of respiratory failure. It is
important to recognize that this weakness may occur in the absence of
meningitis, encephalitis, or even fever or headache—there may be few
other clues that the weakness is due to West Nile Virus infection.
The natural reservoir
of the virus is birds and the virus is
transmitted by mosquitoes (most likely by Culex pipiens, in the North of the US and
Culex quinquefasciatus, in the South; however, West Nile
virus had been detected in many North American mosquito
species). The first human cases in the United States were in 1999 in
New York but in 2002, there were 4156 reported cases of West Nile
virus human infection including 284 deaths, with the virus reaching 44
states. In 2003, it spread into all parts of the United States except
Oregon, Washington and Idaho and in 2004 it was found in all states
except Washington.
The peak incidence of human West Nile virus disease is in late summer
(late August and early September). The spread of the
virus is likely to be due to migration patterns of birds.
In 2003, the number of human cases of the disease was much higher
in western states than in the eastern states in which the
disease was found predominantly in earlier years; for example,
Colorado reported over 1500 cases while South Carolina has
reported only one. This is probably because the bird population
in eastern states has developed immunity against the virus.
It is also probable that West Nile virus has
been transmitted to humans in blood transfusions, organ transplants
and during pregnancy, delivery and breast feeding (vertical
transmission) but these cases are extremely rare.
Treatment for West Nile virus infection is
supportive although it has been suggested
that ribavirin may be active against the virus. In an outbreak in
Israel, however, patients treated with ribavirin had a higher
mortality than those not treated, although this could have been due to
other factors. There is no vaccine.
The best control for West Nile is reducing the
incidence of mosquitoes by spraying (methoprene) or bacterial
larvicides such as Bacillus thuringiensis var. israelensis and
Bacillus sphaericus. Reduction of mosquito breeding sites such
as pools of stagnant water is useful, as is the use of DEET-containing
insect repellents.
|
|
|
|
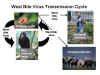 West Nile virus transmission cycle CDC
West Nile virus transmission cycle CDC
|
 A
A
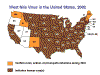 B
B
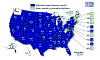 C
C
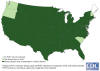 D
D
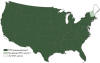 E
E
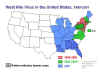
States reporting confirmed West Nile virus infection in
birds, mosquitoes, animals, or humans.
A: 1999-2001 B: 2002 C: 2004 D: 2011 E: 2014 CDC
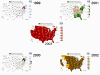 Spread of West Nile Virus in the United States CDC
Spread of West Nile Virus in the United States CDC
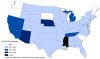 A
A
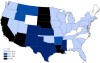 B
B
West Nile virus neuroinvasive disease incidence, by state, United
States, A: 2011 B: 2014
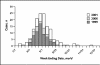 Week of symptom onset for persons
reported to have West Nile virus infection, 1999–2001.
Annals of Internal Medicine
Week of symptom onset for persons
reported to have West Nile virus infection, 1999–2001.
Annals of Internal Medicine
 West Nile virus is a flavivirus commonly found in Africa,
West Asia, the Middle East. and, more recently, in North America. It is closely related to St. Louis encephalitis virus found in the United States. CDC/Cynthia Goldsmith
West Nile virus is a flavivirus commonly found in Africa,
West Asia, the Middle East. and, more recently, in North America. It is closely related to St. Louis encephalitis virus found in the United States. CDC/Cynthia Goldsmith
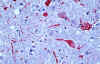
Brain tissue from a West Nile encephalitis patient, showing antigen-positive neurons and neuronal processes (in red)
CDC/W.-J. Shieh and S. Zaki
 US national case definition
US national case definition
|
 Culex quinquefasciatus Mosquito on a Human Finger
CDC
Culex quinquefasciatus Mosquito on a Human Finger
CDC
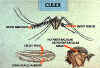 Genus Culex. Note the distinguishing features of the Culex mosquitoes: cross veins on
narrow wings, blunt abdomen, short palpus, and no prespiracular or postspiracular
setae CDC
Genus Culex. Note the distinguishing features of the Culex mosquitoes: cross veins on
narrow wings, blunt abdomen, short palpus, and no prespiracular or postspiracular
setae CDC
|
 Symptoms of West Nile infection.
Annals of Internal Medicine
Symptoms of West Nile infection.
Annals of Internal Medicine |
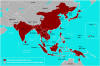 Japanese encephalitis Risk Areas
Japanese encephalitis Risk Areas
CDC |
Japanese Encephalitis
Japanese Encephalitis Virus -
Flavivirus Family
Japanese encephalitis is a mosquito-borne (Culex
tritaeniorhynchus and other Culex species) disease spread
by Japanese Encephalitis (JE) Virus. It is not found in North
America but travelers to rural areas of south and east Asia may
become infected. In temperate areas, the level of infected
mosquitoes and JE virus disease is highest in the summer months but
in tropical areas, highest incidence is associated with the rainy
season.
JE virus infects pigs and birds while humans are "dead-end
hosts".
Most people who are infected by JE virus show no symptoms but in
1% of infected people severe symptoms may arise after an incubation
time of five days to two weeks.
The initial symptoms are:
These are followed by neurological symptoms:
- weakness
- movement disorders
- seizures, usually in children
When the infection causes encephalitis, the mortality rate is 20
- 30% and 30% of survivors will continue to have neurological and
psychiatric problems.
Treatment is supportive but there is a good vaccine which should
be considered by people visiting areas where JE virus is endemic. It
is licensed in the United States and may be given to people over the
age of 2 months.
|
| |
|
ARBOVIRUSES
FEVER AND HEMORRHAGIC FEVER |
|
NAME
|
DISEASE
|
OCCURRENCE
|
VECTOR
|
|
Flaviviridae Family
|
|
Dengue Fever
|
fever, hemorrhagic fever
|
Worldwide - tropic regions
|
Mosquito
|
|
Yellow Fever |
hemorrhagic fever |
South and Central America and Africa |
Mosquito |
|
Zika
|
fever, also causes microcephaly in fetus
|
Africa, Asia, South and Central America. Spreading into
North America
|
Mosquito
|
|
Reoviridae Family
|
|
Colorado tick fever
|
fever
|
North America
|
Tick
|
|
Alphaviridae Family |
|
Chikungunya fever |
fever
encephalitis (rare) |
Worldwide in tropical areas |
Mosquito |
|
Bunyaviridae |
|
Heartland Virus |
fever with Thrombocytopenia |
Missouri and Tennessee |
Tick |
|
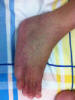
Rash from Chikungunya infection
Wikipedia. Used under Creative Commons

Chikungunya virus
Wikipedia. Used under Creative Commons
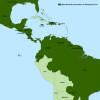
Local transmission of Chikungunya in the Americas
CDC
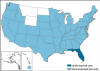
States in the US in which Chikungunya has been
identified. Local transmission from mosquitoes to humans has only be
observed in Florida
CDC
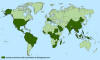
Countries and territories where chikungunya cases have been reported*
(as of October 20, 2015)
*Does not include countries or territories where only imported cases
have been documented.
CDC
|
ARBOVIRUS DISEASES ASSOCIATED WITH FEVER
Colorado Tick Fever
Colorado Tick Fever Virus -
Reovirus
family
This occurs in the Rocky
Mountain States (at elevations above 4000 feet). It is a mild disease resulting
in:
after an incubation period of 1 day to two weeks. Fatigue may
last for a few weeks after the other symptoms resolve.
In very rare cases, there may be central nervous system
involvement resulting in confusion; however, the disease is rarely
life-threatening.
The virus is transmitted
by ticks and is mostly seen in the summer when contact with ticks is most common. In diagnosis, the physician must consider the much more serious Rocky Mountain spotted
fever (rickettsial disease) which may have similar initial symptoms and should
be treated promptly. See also
Ticks
There is no specific treatment other than supportive care.
Chikungunya Fever
Chikungunya Virus - Alphavirus family
Chikungunya virus is a single, positive strand RNA virus in the Alphavirus
family of the Togaviridae. It has spread rapidly around the world. Chikungunya
means that which bends up in the East African Makonde language,
describing the contorted posture assumed by people affected as a result of the
severe joint pain and arthritic symptoms associated with this disease. The
disease was first described in 1955, as a result of an outbreak in 1952 on the
Makonde Plateau, near the border between Mozambique and Tanzania.
Chikungunya virus fever is usually found in warmer regions of Africa and Asia
with some incidence in Europe and other areas imported from endemic areas. In
2013, transmission from mosquitoes to humans (local transmission) was seen in
the Caribbean. In 2014, Chikungunya cases were reported in the United States in
people returning from the Caribbean and by 2015, local transmission from
mosquitoes to humans had been observed in central America, northern South
America and the United States. Most people in the Americans are not immune to
the virus since it has only arrived recently. Thus, infections are expected to
increase and to spread geographically.
Transmission
The vectors for Chikungunya virus are two mosquito species:
Aedes
aegypti and Aedes albopictus. The virus can also be spread by
infected blood and rarely in utero. The newborn can also be infected from
the mother at the time of delivery.
Symptoms
About 3 to 7 days after infection, most patients experience:
Other
symptoms include:
-
headache
-
myalgia
-
conjunctivitis
-
arthritis
-
nausea
-
maculopapular rash
In the laboratory,
lymphopenia,
thrombocytopenia, high creatinine and high hepatic transaminases may be observed.
Most symptoms, which are usually mild, resolve within a week although
polyarthralgia may persist. Some patients, however, experience more severe
disease including infants infected at birth, older people and patients with
chronic conditions. Rare complications include relapse of rheumatological
symptoms and joint pain may last for years. Chikungunya infection is rarely
fatal but has occurred in older patients.
Treatment
There is no vaccine and no specific treatment other than supportive care.
Chikungunya can cause a fatal
encephalitis
There was an outbreak of Chikungunya on the island of
Reunion in 2005. In all, over 300,000 people were infected. Although,
symptoms are usually mild, there were rare cases in which the central
nervous system was involved and some were fatal, particularly in newborns
and patients over 65. The overall case rate of encephalitis (24 in all) was
8.6 per 100,000 infected people. In newborns, the rate was 187 per 100,000
and in people over 65, it was 37 per 100,000 The death rate for patients who
contracted Chikungunya-associated encephalitis was 16%. Many surviving
patients also had long term neurological problems.
|
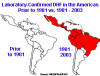
Laboratory-confirmed cases of Dengue Hemorrhagic Fever in the Americas
before 1981 and 1`981-2003
CDC
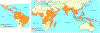
Dengue Fever risk areas around the world
CDC
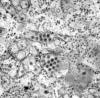
Dengue virus particles. Transmission electron micrograph.
CDC
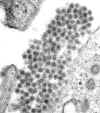
Mature Dengue-2 virus particles replicating in five day old tissue culture cells. The original magnification is 123,000 times.
CDC
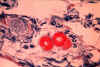 High-power view of heavy fibrin depositis in small arteries, almost occluding the lumen. From a section of
pancreas of a patient who died from dengue hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO)
High-power view of heavy fibrin depositis in small arteries, almost occluding the lumen. From a section of
pancreas of a patient who died from dengue hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO)
|
ARBOVIRUSES ASSOCIATED
WITH HEMORRHAGIC DISEASE
Dengue Fever
Dengue
virus - Flavivirus
family
Dengue fever is the most common arthropod-borne human
disease with over 400 million cases worldwide each year. Transmission is
very rare in the continental United States (the last case was in 2005) but
common in Puerto Rico and other Caribbean islands. Some people have no
symptoms of infection yet are still able to pass on the virus. The virus has
also been transmitted by organ transplants, blood transfusions and possibly
from mother to fetus. WHO estimates that about 22,000 people, mostly
children, die from a Dengue infection each year.
The natural hosts of
Dengue virus are
monkeys and man and mosquitoes (mostly Aedes aegypti but also by Aedes
albopictus) are the vector. Dengue virus has an urban and a jungle cycle.
There are four serotypes of dengue virus (1,2,3,4) and multiple serotypes
circulate in Asia, Africa and the Americas. Dengue is the most common
arthropod-borne human disease and one of
the more rapidly increasing diseases in the tropics. It occurs worldwide
with up to 400 million cases per year. Every year there are cases of dengue fever imported
by travelers into the United States. Usually illness is ~1-8 days after
infection and IgM may not be present until somewhat later.
Symptoms
Acute onset symptoms,
which last about a week, include
-
high grade fever
-
headache
-
rash
-
retro-orbital pain
-
myalgia
-
arthralgia
-
macropapular rash (sometimes)
-
change in taste sensation (sometimes)
-
mild bleeding from nose and gums and easy bruising
-
bone pain (hence the name: "breakbone fever")
In these ways, Dengue may be clinically indistinguishable
from influenza, measles or rubella
The infection can
sometimes progress to encephalitis/encephalopathy.
The symptoms of Dengue are usually milder in children
compared to adults. During the convalescence period there may be prolonged
weakness and anorexia.
Treatment
There is no vaccine and treatment is supportive. The
mortality rate is around 1%.
Dengue Hemorrhagic Fever
Dengue Hemorrhagic Fever is potentially a deadly complication of dengue
and is most severe in children under 15 years of age. Globally, there
are several hundred thousand cases per year.
It appears to be an immunopathological consequence of infection of a person who has already
developed immunity to one serotype of Dengue virus with a virus of another
serotype, although the mechanism is not fully understood. It may well involve an
immune enhancement whereby there is increased uptake of virus coated with
non-neutralizing antibody (developed during an earlier infection with a
different serotype) into macrophages via the Fc receptor. Virus replicates in
macrophages resulting in an increased virus load. In addition, macrophages become
activated and release inflammatory cytokines. The immune enhancement by
non-neutralizing antibody complicates prospects for vaccine development but one
is in development. The disease
is more severe in children; moreover, the presence of maternal antibody in infants may
result in Dengue Hemorrhagic Fever even in a first infection with Dengue virus.
In Dengue Hemorrhagic
Fever, increased vascular permeability is a major problem.
Symptoms
During the initial stages of
Dengue Hemorrhagic Fever, the
symptoms of those of classical Dengue. However, after a week or so, as
the fever subsides, the patients shows:
In most patients this is followed by:
The patients who develop these symptoms are in profound
circulatory shock, in this case known as
Dengue
Shock Syndrome, which can rapidly lead to death.
Signs of Dengue Shock Syndrome are:
Treatment
If the patients with hemorrhagic fever is given suitable
supportive treatment (especially fluid replacement and rest), the mortality rate
is low (2 to 5%) but if left untreated, mortality can be very high (as
high as 50%). The key to survival of the patient is early diagnosis.
CDC recommends that patients thought to have
dengue should be given acetaminophen preparations rather than aspirin,
because the anticoagulant effects of the latter may aggravate the bleeding
tendency associated with dengue infection.
|
|
WEB
RESOURCES
Dengue Fever
CDC
Information - Dengue Fever
|
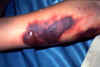 A large subcutaneous hemorrhage on the upper arm of a patient with dengue
hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO)
A large subcutaneous hemorrhage on the upper arm of a patient with dengue
hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO) |
 Aedes albopictus
mosquito feeding. This mosquito is a secondary vector for dengue in
South-east Asia. It recently extended its range into Africa, the New
World and Australia, increasing the risk
of the spread of arbovirus infections. (Image courtesy of the Wellcome Trust)
Aedes albopictus
mosquito feeding. This mosquito is a secondary vector for dengue in
South-east Asia. It recently extended its range into Africa, the New
World and Australia, increasing the risk
of the spread of arbovirus infections. (Image courtesy of the Wellcome Trust)
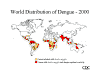 World distribution of dengue fever
CDC
World distribution of dengue fever
CDC
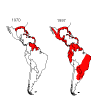 Distribution of Aedes aegypti (red shaded areas) in the Americas in 1970, at the end of the mosquito eradication program, and
in 1997.
CDC
Distribution of Aedes aegypti (red shaded areas) in the Americas in 1970, at the end of the mosquito eradication program, and
in 1997.
CDC
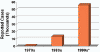 Reported cases of Dengue hemorrhagic fever - 1970's to 1990's
Reported cases of Dengue hemorrhagic fever - 1970's to 1990's
|
|
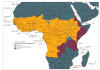 Areas with Risk of Yellow Fever Virus Transmission in Africa. CDC
Areas with Risk of Yellow Fever Virus Transmission in Africa. CDC
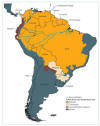 Areas with Risk of Yellow Fever Virus Transmission in South America CDC
Areas with Risk of Yellow Fever Virus Transmission in South America CDC
 Yellow fever virus (magnification: 234,000x) CDC
Yellow fever virus (magnification: 234,000x) CDC

Transmission electron micrograph of Zika virus, which is a member of the
family Flaviviridae. Virus particles are 40 nm in diameter, with
an outer envelope, and an inner dense core. The arrow identifies a
single virus particle.
CDC
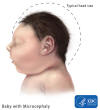
Microcephaly
CDC
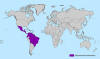
Location of known Zika virus infections: Early 2016
CDC |
Yellow Fever (Hemorrhagic
Fever)
Yellow Fever Virus - Flavivirus
family
This is a disease that is
only found in Africa and South America. In South America, the disease is
sporadic and occurs in forested areas. In Africa, yellow fever occurs mostly
in the rainy season in the west and central areas of the continent. This disease is transmitted by mosquitoes. Natural hosts of the virus include
monkeys and man. It has an urban and a sylvatic cycle.
Symptoms
Most people infected with yellow fever virus have no
illness or only mild illness but some people develop symptoms, after an
incubation period that is typically 3–6 days. Mild illness includes:
-
fever
-
chills
-
severe headache
-
back pain
-
malaise (fatigue)
-
nausea
-
vomiting
Most patients improve after the initial presentation.
However, after a brief remission (hours to a day), about 15% of patients
develop more severe symptoms. This
severe systemic disease is characterized by hemorrhages, degeneration of
the liver, kidney and heart and failure of multiple organs. The
case-fatality rate can be 50%.
Treatment
There is no specific
treatment but there is an effective vaccine
(attenuated strain called 17D). There are sometimes mild effects (head ache,
malaise) of the vaccine within days of administration in a few recipients
(less than 5%) but there have been reports of severe illness (fever, hepatitis
and multiple organ failure) in seven patients in the past six years.
OTHER ARBOVIRUSES
Heartland Virus - Bunyavirus family
Severe Fever
with Thrombocytopenia Syndrome Virus
Heartland virus which is a phlebovirus, a member of the Bunyavididae,
was discovered in 2009 when two farmers from northwestern Missouri became
ill with fever, fatigue, diarrhea, and low levels of blood platelets and
white blood cells. Both men had been bitten by ticks and were hospitalized.
One had malaise after two years while the other initially had fatigue and
memory problems. It is not known if the ticks and the presence of the virus
were related as ticks harboring Heartland virus have not been detected.
As of March 2014, eight cases of Heartland virus disease have been
identified among residents of Missouri and Tennessee. It is unknown at this
time if the virus may be found in other areas of the United States.
Zika Virus -
Flavivirus Family Zika virus, named after the place in Uganda (Zika Forest) where it was
initially identified in 1947, is a small icosahedral positive strand RNA
virus in the Flaviviridae family that has caused recent widespread
alarm. There have been small outbreaks of Zika virus infections for decades
in Africa and Asia. The virus spread to Micronesia in the western pacific in
2007 and then to French Polynesia in 2013. In late 2014, an outbreak of Zika
virus infections occurred first in Easter Island and then on the South
American mainland, particularly Brazil. The occurrence of Zika virus in
Brazil was correlated with a large increase in infant
microcephaly in which an infant is born with an abnormally small head
and an incompletely developed brain. It is thought that infection of the
pregnant mother with the virus is the cause of the abnormal fetal
development. In addition, Zika virus infection may result in some cases of Guillain-Barré syndrome
in adults.
In the United States, up until April 2017, there have been 5,234 cases of
Zika virus disease, almost all of which affected travelers who contracted
the disease in other countries. There have been 223 cases that appear to
have been contracted locally in Florida and Texas as a result of mosquito
bites and 76 cases contracted by other means such as sexual intercourse and
congenital infection. Many more cases have been identified in United
States territories, particularly Puerto Rico. Almost all of these have been
from local mosquito-borne transmission and less than 1% in returning
travelers.
Symptoms
Most people do not show any symptoms of a Zika
virus infection but about 20% will experience some mild symptoms which
may last up to a week. These include:
-
Exanthum (rash)
-
Fever
-
Conjunctivitis
-
Headache
-
Arthralgia
-
Myalgia
Diagnosis
There are specialized tests available to
distinguish Zika from other viral infections that cause similar symptoms
(including Dengue and Chikungunya, see above). These tests include
reverse transcriptase-PCR which detects viral RNA in the blood. There
are also antibody tests to detect anti-viral
IgM. Neutralizing
antibodies usually arise about a week after infection. People who
experience symptoms and have traveled to an area in which Zika outbreaks
have occurred (particular South and Central America) should consult a
health care provider. The virus usually persists in the blood for a week
or more and can be spread by mosquitoes to another person.
Transmission
Zika virus, like Dengue and Chikungunya, is mostly
transmitted by mosquito bites usually by Aedes species mosquito (Aedes
aegypti and Aedes albopictus). Therefore, it is likely to
spread to all areas of North America where these mosquitoes are found.
There are other, rarer routes of transmission:
-
In 2011 in Senegal and 2013 in Tahiti, there
were cases of male to female sexual transmission of Zika virus.
There have also been several reports of sexual transmission in the
United States in 2016. In the case of sexual transmission in Tahiti,
semen and urine of the infected male contained the virus but it was
not detected in his blood. Zika virus has also been found in saliva
although whether it can be spread by kissing is debatable.
-
The virus can be spread by blood transmission
from an infected person.
-
Mother to child transmission has occurred. This
is rare and probably occurs at the time of delivery. There is no
evidence at present for transmission via breast milk.
WHO estimates that up to 4 million people may
become infected by the virus
Treatment
Treatment of the mild symptoms is supportive
including drinking fluids to prevent dehydration and using an analgesic
such as acetaminophen. Non-steroidal anti-inflammatory drugs and aspirin
should not be taken. The best way of avoiding a Zika infection is to
avoid mosquito bites. This can be done by using body-covering clothing,
wearing permethrin-treated clothes, sleeping under mosquito nets or
using insect repellents (such as
DEET). Probably the most effective
short term response to the wave of Zika virus infections will be
mosquito control. There is no anti-Zika vaccine, although it is hoped
that there might be one in a few years. Another possible avenue of
treatment of pregnant mothers is monoclonal anti-Zika antibodies which
will take a much shorter time to develop than a vaccine.
Viral range
Zika outbreaks have been reported from some African
countries, Southeast Asia and some Pacific Islands. In 2015, Zika
infections were reported from Brazil, possibly arriving with infected
persons attending the World Cup. The virus has spread to other
South American countries, Central America, Mexico and the Caribbean
(including Puerto Rico and the US Virgin Islands). It has also been
reported from Cape Verde, an island of the northwest African coast.
Cases have been found in the United States in
people returning from South America. It is likely that the virus will
spread to all areas of the United States where the mosquito vectors are
present.
Microcephaly
Although most people experience no or mild symptoms
of a Zika infection, there is considerable concern about the effect of
the infection during pregnancy. There have been many reports of an
increase in microcephaly in South American countries and these have
coincided with the Zika virus epidemic. Originally, only a small number
of microcephaly cases showed identifiable Zika virus infection; however,
more recent studies using RT-PCR showed that in most, if not all, cases of microcephaly,
all of the infants were infected by the virus. Nevertheless, several
other viruses (including Cytomegalovirus and Chikungunya) can result in
aberrant brain and cranial development.
Microcephaly (small head) is a rare condition. In
Brazil, prior to 2015, only a few hundred cases of microcephaly were
reported each year but since the Zika virus outbreak began there have
been thousands of cases. Some, indeed perhaps the majority, of these
cases may be false positives since there are other reasons that children
are born with small heads, including as noted above, other viral
infections. However, it is clear that infants infected in utero by Zika
do develop microcephaly. Unfortunately, it now appears that
complications in the infant can occur if the pregnant mother is infected
at any time in the pregnancy. Some fetuses have died when mothers were
infected late in pregnancy (25 - 32 weeks) and exhibited brain
calcifications.
Microcephalic infants, besides having a small head and underdeveloped
brain can exhibit seizures, loss of vision and blindness, loss of
hearing, poor balance, learning disabilities and developmental delays.
Microcephaly can be diagnosed in utero using ultrasound. It cannot be cured and
there is no standard protocol for treatment. Some babies with a mild
form of microcephaly have no problems; however, for many there will be
lifelong disabilities, including blindness.
How infection by Zika virus leads to brain
malformation in the fetus is not known but the virus seems to destroy
cortico neural progenitor cells that will form the brain cortex.
In the United States, the incidence of microcephaly
is 2 – 12 per 10,000 live births (CDC figures)
Miscarriage
Zika also appears to result in miscarriages.
Because of this, CDC has advised pregnant women not to travel to
countries where Zika infection is a risk and El Salvador has suggested
that because of the outbreak, potential mothers should delay their
pregnancies (which may be difficult as birth control is not widely
available).
Guillain-Barré syndrome
Guillain-Barré syndrome is a rare autoimmune
disease in which nerve cells become damaged, leading sometimes to
paralysis and death. In the United States, there are between 3,000 and
6,000 cases per year. Most patients, however, completely recover. It is
not known what causes Guillain-Barré syndrome but it often follows a
viral or bacterial infection. It is not established that an infection by
Zika virus can cause Guillain-Barré syndrome since it is always
difficult to find any cause in a particular patient. Rather there are
correlations and in Brazil there has been an increase in Guillain-Barré
syndrome at the same time as the rise in Zika virus infections.
|
|
WEB
RESOURCES
Yellow Fever
CDC
Information Chikungunya
CDC
Information Zika
CDC Information |
|
 Return to the Virology section of Microbiology and Immunology On-line
Return to the Virology section of Microbiology and Immunology On-line
This page last changed on
Saturday, April 15, 2017
Page maintained by
Richard Hunt
|

 Components
in the transmission and maintenance of Arboviral Encephalitis
CDC
Components
in the transmission and maintenance of Arboviral Encephalitis
CDC Sylvatic life cycle
Sylvatic life cycle
 St Louis encephalitis virus transmission
cycle CDC
St Louis encephalitis virus transmission
cycle CDC A
A  A
A Culex quinquefasciatus Mosquito on a Human Finger
CDC
Culex quinquefasciatus Mosquito on a Human Finger
CDC Symptoms of West Nile infection.
Annals of Internal Medicine
Symptoms of West Nile infection.
Annals of Internal Medicine Japanese encephalitis Risk Areas
Japanese encephalitis Risk Areas

 A large subcutaneous hemorrhage on the upper arm of a patient with dengue
hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO)
A large subcutaneous hemorrhage on the upper arm of a patient with dengue
hemorrhagic fever. (Image courtesy of the Wellcome
Trust/WHO)
















































